Decoding the FTO genetic puzzle
The story of scientists trying to crack open a low-hanging fruit they picked many years ago
Happy Friday! For this week’s post, I chose one of my most popular Twitter stories that I wrote last year. This is a great example of to illustrate the challenges of using common variants to do drug target discovery. In a long road of genetics-driven drug development, ideally the starting point is a gene. Rare variants get us straight on the road, but the common variants have a tendency to lead us astray. And that is why drug developers go after rare variants for target discovery.
From the Twitter archives
Buckle up! We're in for a wild ride today. A paper in Nature Metabolism by scientists from China adds a surprising twist to the long-known FTO GWAS story.
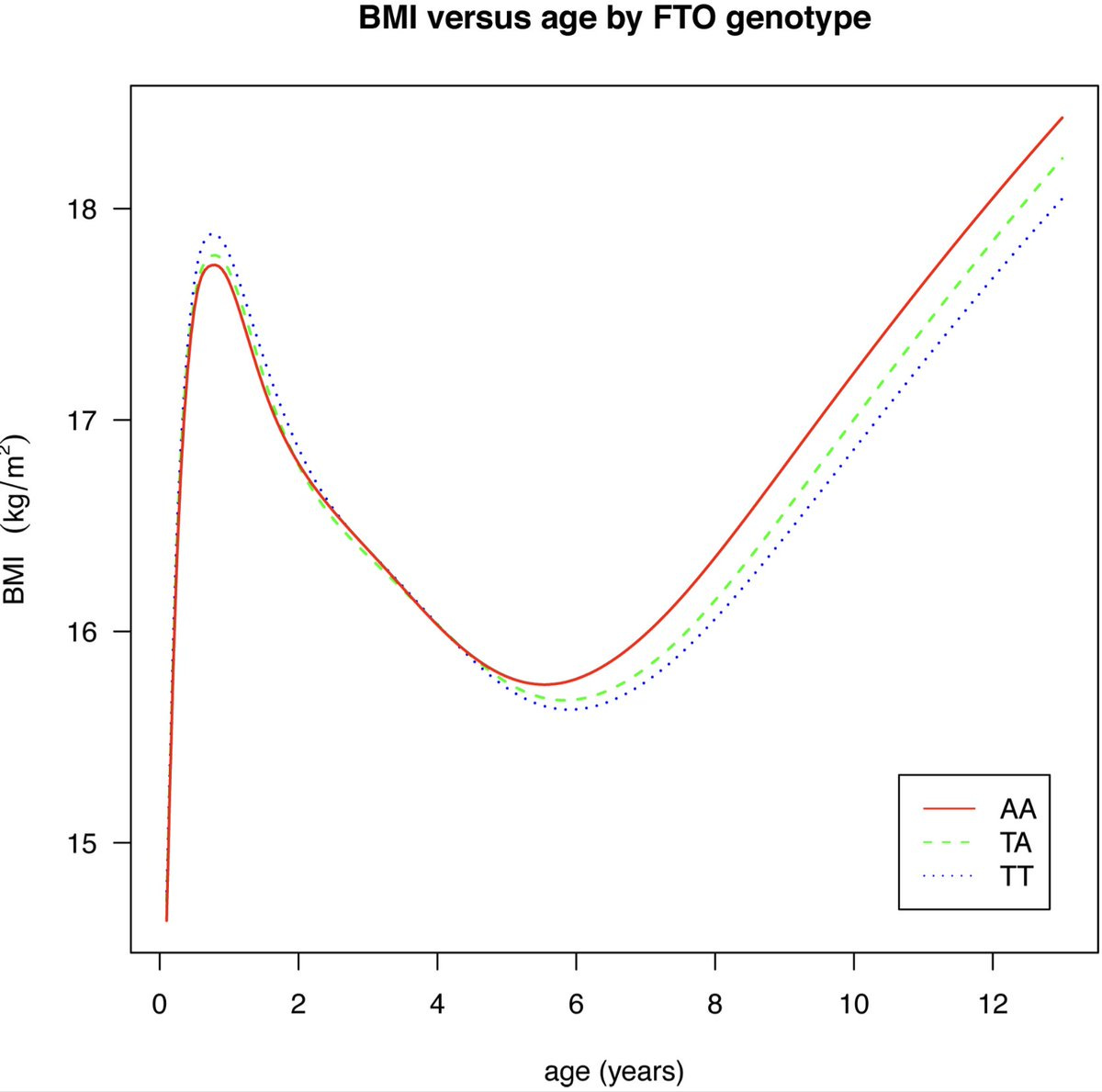
The FTO locus (16q12.2) is the first ever GWAS locus to be associated with obesity and even after 16 yrs now, scientists appear to be scratching their heads trying to make sense of this locus. Non-coding intronic variants within FTO strongly associate with BMI, where individuals homozygous for the top risk variant weigh ~3kg more than non-carriers.
Since its discovery in 2007, there have been tremendous efforts to identify the causal gene(s) at this locus. Given that the risk variants are sitting right within a gene, FTO was of course the primary suspect. How do you find out if FTO has an effect on BMI? Delete it in mice and see if the animal gains weight. And that's what scientists did and found out that Fto knockout mice were stunted and lean, and the leanness was mainly due to burning too much fat. That's great. So FTO must be the causal gene.
But then contradicting findings appeared. If you knock out the Fto only in adipose tissue or globally after the animal has grown, the mice actually gain weight!
Amidst this confusion came an even bigger one: two landmark papers, one in Nature and the other in NEJM, said, forget about FTO, the causal genes are located far away. The FTO locus is an enhancer that folds in the 3d space and touches the promoter of distant genes IRX3 and IRX5. And deleting Irx3 in mice resulted in weight loss.
Then scientists were like, you know, a proper experiment would be not to knock out Fto or Irx3, but to delete the homologous noncoding region in mice. So, they deleted an 82 base pair-homologous region in mice and showed that without this region, the mice don't gain weight when fed with a high-fat diet, and deleting this locus increases Irx3 and Irx4 expression. So, the causal genes are IRX3 and IRX4 then.
And now, in the current paper, the scientists argue, you know, the most appropriate way to study the FTO locus is to recreate the exact genetic variant in mice and study the consequences.
What did they find? The exact opposite of what was found in humans. The risk allele, that increased weight in humans, decreased weight in mice. Can it be because of some off-target effects of the CRISPR experiment? No. Even if you do the knock-in in an old-fashioned way, the results are the same. The weight loss is mainly via over energy expenditure via brown adipose tissue. Wait, there is another twist.
There is an interesting difference between humans and mice. Humans have brown adipose tissue only during infancy and then lose it as they grow into adults, which isn't the case in mice. The brown adipose tissue helps mainly during cold temperatures. The experiment mice are usually housed at around 20-22 degrees Celsius, which is an ambient temperature for humans but not for mice. It's substantially cooler than mice's "thermoneutral zone (29-31 degrees)".
So what happens when you repeat the experiments in what might be the mice's ambient temperature--30 degrees? All the weight loss effects that were previously seen at 20–22 degrees are now blunted. So, the FTO locus effects are strongly dependent on two things: temperature and the presence of brown adipose tissue. And it turned out, in fact, it was previously shown in humans that the FTO variant has an age-dependent effect. It lowers the body weight in infants and then increases the body weight in adults, which aligns with the current finding.
Overall, the current paper is an impressive work and will stand as a landmark in the long twisty road of FTO story. But above all, this paper is a remarkable example to show case the challenges behind translating mice physiology to human physiology.