GWAS stories: Thanksgiving edition
The fascinating genetics of smelling and tasting
The tale of Icelanders who lost their ability to smell rotten fish
I love fish. Particularly when it is coated with cream made of chili powder, coriander powder, garam masala, salt and other Indian herbs and deep fried in oil. A tasty irresistible non-vegetarian side dish popular in the Southern states of India.
I still remember the day in 2015 when I was at a Christmas dinner event organized by the department of Biomedicine at Aarhus University in Denmark. I have just started living in Denmark, and still, brand new to Danish traditions and food. One of the dishes served was pickled herring, a traditional Danish dish. I had no knowledge of this dish before that day. I imagined that it should be tasty and helped myself with an extra piece, a decision that I regretted the moment I had my first bite. It took me almost 30 seconds to push that bite inside me without evoking my gag reflex. My colleague who was sitting next to me informed me that what I am staring at with absolute disgust is fermented fish, a traditional Danish dish that everyone loves to eat, particularly during Christmas. As a newbie, I need to acquire the taste through experience to enjoy the dish, he added. I lived in Denmark for the next 5 years, and I never managed to grow the taste buds for fermented fish. Every time I attempted to taste it, I only disliked it more.
A fascinating paper was published in 2020 by the deCODE scientists. It got substantial press coverage. New York Times published an article about the findings. In the paper, the authors report a GWAS of odor perception conducted in around 11,000 Icelanders. The participants were asked to sniff six different odors—licorice, cinnamon, fish, lemon, peppermint, and banana—and name them and report on the intensity and pleasantness of the odor they perceived. Analyzing the associations of nearly 35 million genetic variants with various odor perceptions, the authors uncovered three genome-wide significant loci of which one, located in chromosome 6, was associated with fish odor perception. At this locus, a missense variant in TAAR5, which encodes an olfactory receptor, reduced the intensity of fish odor perception by 0.37 SD, an effect strong enough to say the carriers are blissfully unaware of the foul-smelling characteristic of the odor caused by a volatile chemical called trimethylamine (TMA) that is prototypical of fish odor. The carriers of this missense variant described the fish odor using terms such as “ketchup”, “caramel” or “rose” (seriously, rose?). Few of the carriers of this missense variant were homozygotes (likely a knockout) and their perception of the fish odor turned out to be even worse, suggesting a dose-dependent effect.
The gene TAAR5 encodes a member of the TAAR family of trace-amine-associated receptors expressed in the olfactory epithelium of vertebrates and are essential for detecting aversive amines such as TMA (the one in the odor that participants sniffed), which “is a bacterial metabolite found in rotten fish, animal odors, and human bodily secretions such as sweat, bad breath, urine, feces, and blood.” The identified missense variant is common in Europeans but rare in non-Europeans. The risk allele frequency follows a North-South gradient, ranging from ~2.2% in Icelanders (the highest) to 0.2% in Africans. The TAAR5 receptor seems to be activated specifically by rotten salmon but not by fresh salmon, and the evolutionary importance of this receptor might be that it protected humans and other vertebrates from ingesting spoiled foods loaded with harmful pathogens. But fascinatingly, the loss of this receptor function seemed to have benefited the Europeans, particularly the Northern Europeans such as Icelanders whose diet is characterized by “extraordinarily high consumption of fish”. It is a common practice in Iceland and other Scandinavian countries, especially during the Middle Ages, to eat fermented fish because of the scarcity of fresh fish during the harsh winters. The authors write “Fermented skate is considered a delicacy by many and eaten on traditional occasions by around 35% of the population, despite its aversive odor.“ Hence, it’s likely that this variant underwent a positive selection in Iceland and other European countries.
Under normal circumstances, the fact that there are millions of humans in Europe who can’t smell rotten fish would have blown my mind away. But having lived in Denmark for more than 5 years and watched people immensely enjoy eating raw fermented fish, I wasn’t surprised at all.
An interesting example of a natural event boosting the genetic signal
When the pandemic hit, I was in Denmark, working as a postdoctoral researcher at Aarhus University. I moved to New York in May 2021 to start a new job as a research scientist at the Regeneron Genetics Center. Somehow I evaded COVID-19 until the end of 2021. I had my vaccinations up to date. Despite that, COVID-19 hit me in January 2022 when I was visiting my parents in India. I guess it was the omicron, the prevalent strain at the time. I had most of the typical symptoms except one: loss of smell and taste. Despite the severe cough and myalgia, I continued to enjoy Indian cuisine, something that I can only do so while in India: filter coffee, masala-dosa, poori, idiyappam, appam, thalapakatti biriyani, the list goes on. (I am already salivating as I type these words, sitting in my apartment in New York). If you aren’t familiar with the dishes that I just listed, it’s okay. They are not important for the story. So, what I am trying to say is after getting the infection, I learned that I am not one of those people whose main COVID-19 symptom is loss of smell and taste.
Scientists from the 23andme published a fascinating GWAS, and the timing of the publication couldn’t be any more perfect: just three days before I tested positive for COVID-19, by when I was already sick and knew it was covid. I remembered reading the article while I was at the peak of the infection and quarantined at my home. The findings in the paper inspired me to frequently test my sense of smell and taste. I smelled everything that was served to me.
In the paper, the authors report a GWAS locus that they found to increase the odds of experiencing loss of smell or taste when infected with COVID-19 (OR=1.11; P=1.4e-14). The locus was near genes UGTA1 and UGTA2 that encode members of a family of uridine diphosphate glycosyltransferases, enzymes that convert fat-soluble metabolites into water-soluble ones by conjugating glucuronic acid. We have learned about these enzymes mainly in the context of bilirubin and xenobiotics metabolism in the liver. But it turns out that these enzymes are also expressed in the olfactory epithelium and help get rid of unwanted odorants by conjugating them with glucuronic acid, chemical change that prevents the odorants from binding to olfactory receptors.

The fact that most fascinated me is that this locus was not COVID-19-specific but it was also associated with the general ability to smell. Since COVID-19 infection affects the taste and smell perception, it’s likely that, by studying only COVID-19-infected individuals, the authors have simply boosted the statistical power to identify a GWAS locus associated with the general ability to smell as, in the authors’ own words, “those with heightened smell or taste sensitivity may be more prone to notice a loss of these senses resulting from a SARS-CoV-2 infection.”
I like this finding because it is a beautiful example of a gene-by-environment interaction. And it teaches us that one can significantly increase the statistical power to detect a genetic signal by leveraging natural events such as COVID-19 infection. If the authors have to detect the same signal in the general population by asking them how good they are with their smell and taste perceptions, they will probably need a much bigger sample size.
A Mendelian human trait of odor perception
Human diseases and traits can be monogenic, where the disease or trait variation is caused by a single gene mutation (e.g. cystic fibrosis), or polygenic, where the disease or trait variation is caused by hundreds to thousands of variants, each increasing the odds of disease or modifying the trait variation by a minuscule percentage (e.g. depression, height). Note, diseases that are typically polygenic can sometimes take monogenic forms, a very good example is the monogenic form of obesity caused by MC4R mutations. Human diseases and traits can also sometimes lie in between the monogenic and polygenic ends of the spectrum and are termed oligogenic, i.e., the disease or the trait variation in the population can be explained almost fully by only a handful of genetic variations spread over a small proportion of the genome as opposed to thousands spread over the entire genome. While it’s common for human diseases to be monogenic or oligogenic, it’s rare for human traits to have monogenic or oligogenic causes (e.g. eye color). And it’s even rarer for human traits to have a singular genetic cause where almost the entire phenotypic variation is explained by a single genetic variant. I came across one such trait when reading about the genetics of odor perception.
Our repertoire of odor perception spans hundreds of receptors, and it’s likely that when one gene is lost, another takes over. But sometimes, our ability to perceive a specific odor boils down to a single gene, which seems to be the case for odor from β-ionone, a chemical from the family of rose ketones, an important contributor to the rose fragrance.
A GWAS of variations in sensitivity to various food-related odors identified an extremely strong genome-wide significant locus in chromosome 11 associated with the sensitivity to β-ionone odor. The causal variant at this locus was found to be a missense variant in OR5A1, an odor receptor gene. The variant rs6591536 changes the amino acid aspartic acid (allele G) to asparagine (allele A) at position 183 that maps to an extracellular domain of the protein. It turns out that there is a whopping 100-fold difference in the β-ionone sensitivity between G allele heterozygotes and homozygotes (AG and GG) compared to A allele homozygotes (AA). Asparagine substitution in both the copies of the OR5A1 protein (AA) knocks down the sensitivity to β-ionone by a whopping 100 folds. The difference is so strong that this missense variant alone explains >96% of the phenotypic variation in our ability to smell rose fragrance, making the β-ionone odor perception a Mendelian human trait.
The OR5A1 missense variant is common across all populations and so, can be easily replicated in any human population. Genetic association analysis of beta-ionone sensitivity in a mere 109 Southeast-Asians yielded an impressive P value of 1.5e-22. A Plos genetics paper published early this year (2022) reports a GWAS of odor sensitivity in 1000 Han Chinese and impressively replicates the association of the OR5A1 missense variant association with β-ionone.
It’s absolutely fascinating to imagine that a random mutation that happened sometime perhaps hundreds of thousands years ago divided entire humanity into two groups: one who can easily smell the rose fragrance and the other who can’t. If we measure the β-ionone sensitivity as a quantitative trait in a random sample of humans from anywhere in the world and plot the results, the values will almost always follow a bimodal distribution. Such a large effect variant obviously will impact human behavior in terms of making choices about food and fragrance. The extremely high sensitivity to the odor doesn’t always translate into a pleasant experience, and the behavior can very much depend on the context. In an interesting set of behavioral experiments, Jaeger et al. show that G allele carriers (AG and GG) with high sensitivity avoided chocolates and apple juices that are flavored with β-ionone while A allele homozygotes didn’t. At the same time, the G allele carriers preferred non-food items such as household fragrance products more when they contained β-ionone; AA genotype carriers, however, didn’t exhibit any such preference. What a beautiful example of a human genetic variation influencing human behavior!
So, next time when you are with your friends at a flower shop or a perfume shop, find out how many of you can smell the rose fragrance easily and how many of you can’t. The ones who can’t are likely homozygous for A allele of the OR5A1 missense variant.
FGF21—the sweet tooth gene
How can I finish a Thanksgiving edition without dessert? Let’s talk about FGF21, a sugar-induced hormone that influences one’s tendency to like sweet food.
I have sweet tooth. I love desserts and sweets. Although I wasn’t a great fan of Danish cuisine, I absolutely loved Danish pastries. Danes add so much sugar to their pastries, and it compensated me well for the lack of tasty and spicy food in Danish cuisine. My favorite Danish pastry is “Brunsviger“, a delicious Danish cake with a soft and moist dough coated with a thick layer of melted brown sugar. Brunsviger is a popular cake in Denmark and has been in existence for many generations.

I travelled to Aarhus for a couple of days this year (2022) September after attending the World Congress of Psychiatric Genetics (WCPG) in Florence, Italy. One of the items on my TODO list was to visit Lagkagehuset, a popular Danish bakery located close to where I used to live. Unfortunately, Brunsviger was sold out when I visited the shop, and I felt really disappointed. Still, I enjoyed the other tasty cakes and desserts.
Everyone is aware of the hormone insulin, which is secreted by the beta cells of the pancreas in response to blood glucose levels. Obviously, it is came to your mind when I said sweet hormone, isn’t it? But do you know that there is also another insulin-like hormone called FGF21 that spikes in our blood when we binge on sweets? Increased level of this hormone is associated with decreased sweet consumption and vice versa. The initial discovery of FGF21 as an insulin-like hormone was made in early 2000 through an in vitro phenotypic screen carried out at Lilly research laboratories. Several novel proteins of unknown function were tested for their effect on glucose uptake in adipocyte cultures, and one of the proteins, FGF21, emerged as highly significant causing increased glucose uptake by the adipocytes. Many animal studies naturally followed this discovery, and today, we know a lot about the FGF21 biology. It’s a hormone under feedback regulation. It comes from the liver, acts on the brain and suppresses our sugar and alcohol cravings, in addition to numerous other actions.
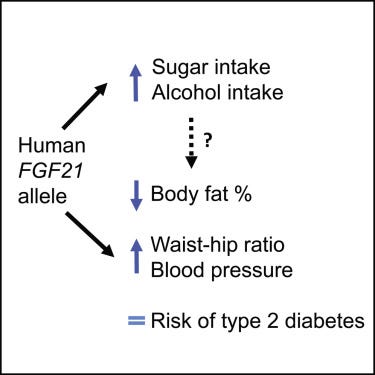
As a human geneticist, the most fascinating part of the FGF21 story for me is the discovery of the GWAS signal at the FGF21 locus associated with both carbohydrate and alcohol consumption. The first discovery of FGF21’s genetic association with sugar intake was made by Danish scientists. (Now you know why I started this story with Danish pastries ;) ) Soberg et al genotyped FGF21 variants in ~6500 Danes and studied the genetic associations with a range of food behaviors. This revealed a significant association between a synonymous FGF21 variant (rs838133) and candy consumption. The authors also found a nominal association with alcohol consumption. Although this was a candidate gene study, the associations with both sweet and alcohol consumption were proved to be real in subsequent GWAS in large sample sizes. This variant is highly pleiotropic with the strongest associations seen with two main phenotypes: carbohydrate and alcohol consumption. The synonymous variant decreases FGF21 levels in the blood and consequently, increases sweet and booze consumption in humans (and also, mice). What is even more fascinating is this variant, despite increasing sweet consumption, is not associated with an increased risk for type 2 diabetes. It’s not clear why, but it might be due to the variant’s effect on other macronutrients intake, for example, the variant is also associated with decreased fat intake.
Today, FGF21 analogs are being tested in preclinical and clinical studies for many metabolic indications including obesity, type 2 diabetes, non-alcoholic fatty liver disease, etc. However, as far as I know, there is no consensus yet on the true therapeutic value of FGF21 analogs. But let’s not worry about that for now. I’ll dive deep into the FGF21 story from a therapeutic angle in one of the future posts.
That’s all for today. I’ll let you get back to your Thanksgiving leftovers. Enjoy the rest of the holidays.
—Veera










Nattō (fermented soybean) is a traditional Japanese breakfast item. I had tasted it once and would never want to smell it again, let alone ingesting it. I wonder if similar findings can (or have been) be made in Japanese population.